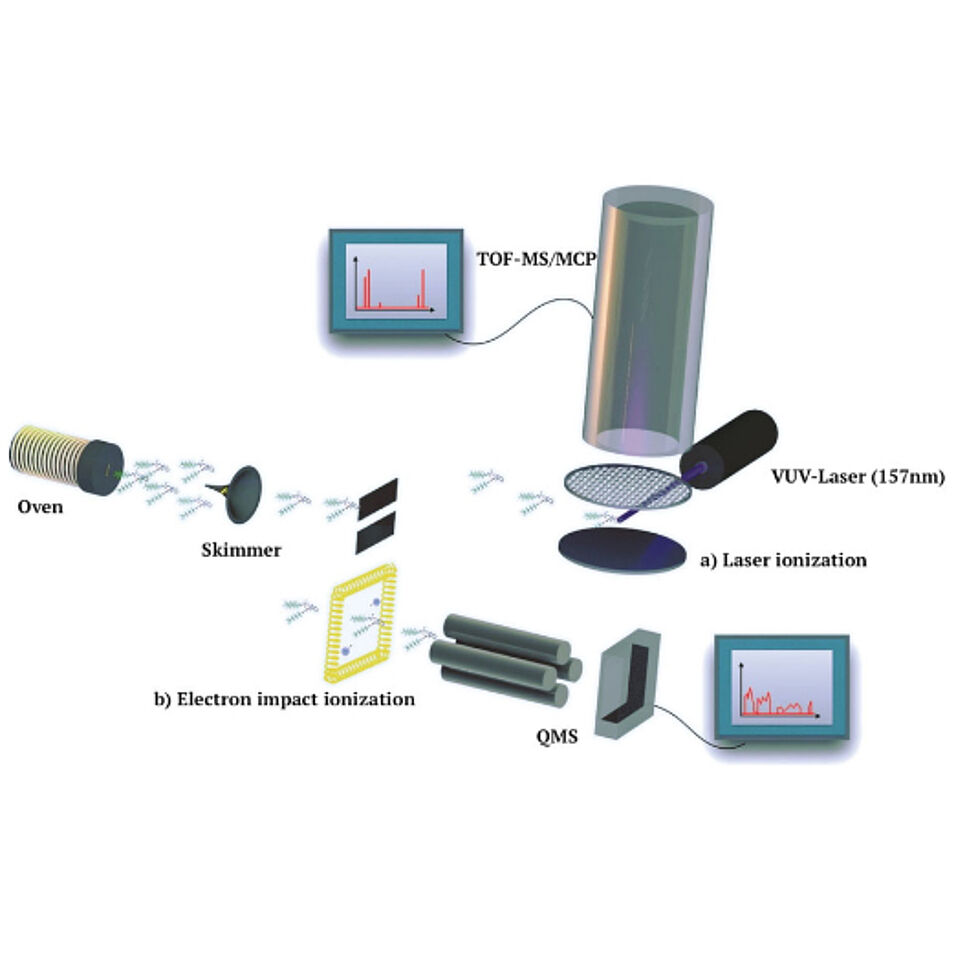
Amino acids are essential building blocks of life, and fluorinated derivatives have gained interest in chemistry and medicine. Modern mass spectrometry has enabled the study of oligo- and polypeptides as isolated entities in the gas phase, but predominantly as singly or even multiply charged species. While laser desorption of neutral peptides into adiabatically expanding supersonic noble gas jets is possible, UV–VIS spectroscopy, electric or magnetic deflectometry as well as quantum interferometry would profit from the possibility to prepare thermally slow molecular beams. This has typically been precluded by the fragility of the peptide bond and the fact that a peptide would rather ‘fry’, i.e. denature and fragment than ‘fly’. Here, we explore how tailored perfluoroalkyl functionalization can reduce the intermolecular binding and thus increase the volatility of peptides and compare it to previously explored methylation, acylation and amidation of peptides. We show that this strategy is essential and enables the formation of thermal beams of intact neutral tripeptides, whereas only fragments were observed for an extensively fluoroalkyl-decorated nonapeptide.
Read the full article here.
J. Schätti, U. Sezer, S. Pedalino, J. P. Cotter, M. Arndt, M. Mayor, V. Köhler,
Tailoring the volatility and stability of oligopeptides,
Journal of Mass Spectrometry 52, 550-556 (2017).